What Makes REDLEAF Spinal Implants Stand Out?
- 35 years of origin manufacturer, long-term cooperation and win-win with you.
- Focused on the research and manufacturing of orthopedic spinal implant systems.
- Used by orthopedic doctors and distributors in multiple countries around the world.
- Professional orthopedic experts and clinical physicians provide technical and design based on the characteristics of human skeletal structure.
- Years of overseas sales experience, providing one-stop surgical solutions.
- Our professional after-sales team is always available online to answer your questions.
Professional Spinal Implants Manufacturer for Global
As a professional factory with over 35 years of experience in orthopedic implant manufacturing, REDLEAF has dedicated orthopedic R&D experts and clinical physicians to provide technical guidance. Each product is designed based on the characteristics of human skeletal structure and has been validated through multiple clinical trials. The professional design of the spinal nail and rod system ensures the stability and safety of the product during surgery.
Each product complies with international medical device standards and accepts clinical customization to meet your various orthopedic implant needs. We can help you solve various clinical surgical scenarios.
Classification of Spinal Implants
Spinal implants are developed based on the structural characteristics of the human spine, forming a rigid structure through multi-point fixation to fix the spine, limit abnormal spinal activity, and reduce spinal segment lesions.

Integrated work channel completes rod placement and screw tightening,Can eliminate disassembly work

Adopting variable pitch thread and plum blossom top thread design, Can be more sturdy, stable, and prevent sliding

Screws and steel plates have a certain adjustable angle fixation in the coronal and sagittal planes
The lumbar and cervical fusion cage is made of Peek biomaterials, which have good biocompatibility

Cannot be used for fractures, tumors, especially severe osteoporosis that have lost sufficient anterior column support

A complete set of surgical tools can not only save surgical time, but also increase the probability of surgical success
Advantages of Spinal Implants System
We have a comprehensive range of spinal implant products, and all products sold undergo rigorous clinical validation to ensure their safety and effectiveness.

Select appropriate implants based on the patient’s spinal condition to achieve precise treatment.

Titanium alloys and Peek biomaterials have excellent biocompatibility, avoiding rejection reactions in the human body

Our products undergo rigorous clinical trials before being launched to meet the diverse clinical needs of physicians.

Our spine product category is comprehensive and can meet the diverse procurement needs of distributors.
Spinal Implant Minimally Invasive Surgery
Spinal implant Minimally invasive surgery is a percutaneous, minimally invasive surgical procedure performed with a minimally invasive spinal screw-rod system.Doctors can use the accompanying surgical instruments to perform multiple percutaneous punctures and implant break-off U-shaped long-tailed pedicle screws, connecting rods, and other spinal implants at the wound site to achieve a stable fixation effect.
Universal pedicle screws (long tail) can complete the implantation and locking through an integrated working channel, avoiding the cumbersome installation and removal of long tail pedicle screws during surgery. This design shortens the operation time, and making it easier to recover from the minimally invasive wound after surgery.

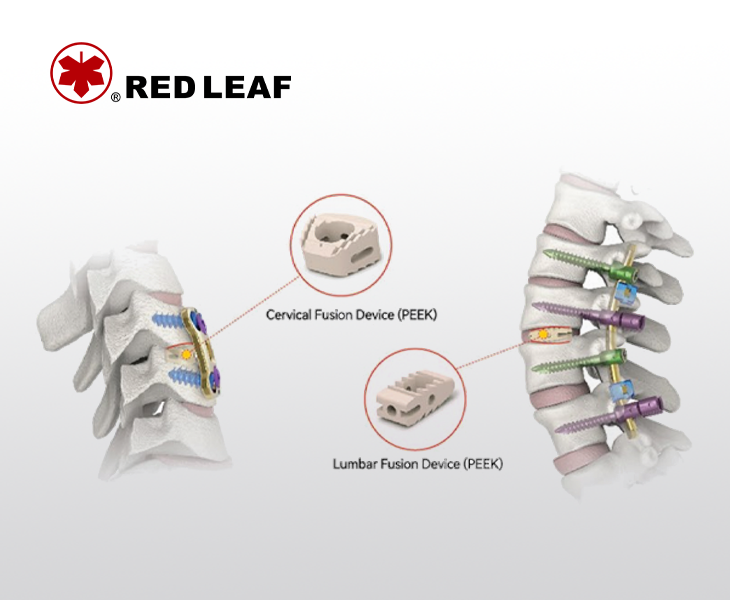
Fusion Device System
Interbody fusion devices system made of polyetheretherketone (PEEK) are indispensable in Spinal implant surgery.Polyetheretherketone (PEEK) possesses excellent biocompatibility, corrosion resistance, high elasticity, and radiolucency, making it a next-generation orthopedic implant material that succeeds titanium alloys. When implanted into the intervertebral space in conjunction with a Spinal implant product, these devices can maintain long-term stability, promote bone fusion, and help address conditions such as vertebral lesions or tumors.
Product Resource Download

This article can describe the technical features of the products, including its working principle and applicability, which can helps you provide some reference for clinical practice.

A video demonstration of the locking plate can visually demonstrate the use of the product and related equipment, showcasing product performance and operation process.

We can provide you with ISO international certification documents, product testing reports, or other documents to assist you with distributor registration and government bidding.
A Trustworthy Orthopedic Manufacturer


We have been deeply involved in the field of orthopedic implants for decades, and our actual industry experience has never been built on slogans. We have professional R&D experts, and all steel plates are designed to fit the anatomical shape of the human body, making them safe and reassuring to use. This is also our confidence in serving you.
All products undergo strict testing from the raw materials entering the factory, followed by CNC precision machining, polishing, cleaning, packaging, and complete quality control at every stage, in order to provide stable, safe, and reliable orthopedic implant products to global customers.
Example of Locking Plate Solution
We are available 24 hours a day and welcome partners from around the world to contact us. Below are some application cases of locking plates in actual surgical processes that we have collected. Please take a look
With the acceleration of global population aging, fractures caused by osteoporosis have become an increasingly common problem in clinical trauma orthopedics. Compared to conventional steel plates, screws are more prone to loosening and fixation failure. The angled stable locking structural design of the locking plate allows for the establishment of a stable healing environment suitable for the wound site without excessive compression of bone soft tissue, preventing screw loosening and the probability of surgical failure at the root.
MIPO technology in orthopedic surgery is a solution that promotes healing by reducing soft tissue damage and preserving bone blood supply. This surgery does not require large incisions, only limited small incisions are needed to percutaneously implant the locking plate, which can protect the soft tissue and periosteum around the fracture to the greatest extent possible.This plan is suitable for reducing the risk of postoperative infections and complications such as bone non union.
Serious injuries, postoperative infections, and other difficult cases of fractures cannot be treated in one step, and can only be treated step by step in stages. Firstly, control the infection, clean the lesion, and then reconstruct and fix the bone defect. In the early stage of reconstruction and repair, locking plate can be of great use, which can firmly maintain the position of the bone and gradually restore the patient’s mobility. It is widely used in major trauma surgeries and reconstruction specialties.
Welcome To Learn About Us
- Factory Photos
- Certificate Image
- Process Flow



Real feedback from global customers
In the treatment of orthopedic trauma, locking plates are commonly used for osteoporotic fractures, complex fractures, and minimally invasive percutaneous fixation in elderly patients. The angled stable structural design of this product can provide optimal stability and help maintain a stable healing environment at the wound site. Especially in the case of insufficient bone mass or more fracture fragments, stable internal fixation support can often speed up the recovery progress after surgery.
Our locking plates are mostly made of medical grade titanium alloy, which has good biocompatibility and corrosion resistance. To ensure that patients do not experience significant adverse reactions after implantation, regular imaging examinations after surgery can also be performed to help doctors timely assess the progress of fracture healing.
Locking steel plate is a type of steel plate that has both regular threaded holes and locking threaded holes. During surgery, regular threaded nails are usually implanted first, and then the locking holes are gradually implanted to support the overall structure.
Usually, when facing comminuted fractures, the steel plate is chosen to be 2-3 times the total length of the fracture. For simple fractures, it is usually 8-10 times the total length of the fracture. This helps to distribute pressure more evenly at the trauma site and reduce postoperative risks
The commonly used orthopedic steel plate materials on the market are generally divided into stainless steel and titanium alloy. Stainless steel material has high strength but low price, and its compatibility in the human body is not high. In comparison, titanium alloy has higher biocompatibility and is easier to be compatible in the human body.
LCDCP usually refers to a locking pressure plate with two different sides, one of which has grooves on the surface, which can minimize the contact area with cartilage tissue during surgery, reduce damage to bone tissue, and facilitate your later recovery
Precision Machining – High Quality Production
In the production process of orthopedic internal fixation, the size and quality of the steel plate often directly affect the success rate of the surgery. As a orthopedic manufacturer with over 30 years of experience, we have introduced DMG MORI’s high-end CNC machining equipment to help our products achieve stable and consistent size and structure of locking steel plate products.
We are also equipped with the TZTEK Tianzhun measurement system, which uses a high-power automatic zoom lens, magnification, and AI to automatically grasp edges, detect key hole positions, plate curvature, and thickness tolerances throughout the entire process, and reduce errors caused by manual intervention. Full process quality control ensures that every steel plate undergoes strict quality control before leaving the factory, reducing the burden on doctors in labor, improving success rates, and providing guarantees for the safety of patients’ lives.
Dust free clean room
A sterile and clean production environment can also directly affect the success of surgery. From precision machining, ultrasonic cleaning to terminal aseptic packaging, our production workshop complies with ISO and GMP quality management standards. Cleaning and packaging are completed in a sterile clean room that meets the 100000 level standard. Each process is carried out in a constant temperature, humidity, and enclosed controlled environment, which helps reduce the possibility of contamination.
In order to ensure 100% quality of product delivery, systematic sterilization validation and quality sampling will be carried out before leaving the factory to ensure that the products meet international medical device registration and clinical use requirements, providing the best product guarantee for each of our customers. This is also our confidence in facing global distributors and related institutions.













